Melius Organics and REAXENSE Partner to Develop a SuFEx Hub-Enabled Platform for Chemical-Space Exploration
REUS, Spain and TORONTO, Canada, April 30th, 2026
Melius Organics and REAXENSE today announced a collaboration to build a Sulfur(VI) Fluoride Exchange (SuFEx) Platform that pairs hub-enabled SuFEx chemistry spanning sulfonyl fluorides, fluorosulfates, sulfamoyl fluorides, and SOF₄-derived aza hubs with AI-assisted library design, exploration, and prioritization. The platform will enable modular, scalable assembly of functionally diverse chemical libraries while guiding compound selection toward synthesis-ready candidates. In practice, this delivers SuFEx-enabled discovery workflows that are faster to deploy, easier to scale, and more predictable, supporting target-focused covalent fragment campaigns, rapid expansion of hits into synthesis-ready analog series, and late-stage functionalization programs across drug discovery and chemical biology.[1,2]
Since Sharpless and colleagues first described it as “another good reaction for click chemistry,” SuFEx has grown into a genuinely modular platform. Its core feature, the formation of highly stable S(VI)–X linkages under mild conditions, now extends across a family of connective hubs with distinct and complementary reactivity profiles.[1]
The first generation of di-oxygenated hubs (sulfonyl fluorides, fluorosulfates, and sulfamoyl fluorides) provides linear connectivity with well-characterized synthesis and reactivity. More recently, SOF₄-derived iminosulfur oxydifluoride intermediates have unlocked a second generation: mono- and di-aza S(VI) hubs, including sulfonimidoyl fluorides, sulfurofluoridoimidates, and sulfuramidimidoyl fluorides (Figure 1). These newer hubs introduce branching architectures and extra diversification handles. In practice, this means chemists can now pick hubs by geometry, electrophilicity, and orthogonality (O-selective vs. N-selective entry points, for instance) to tune the connectivity, stability, and reactivity profile that a given target demands.[1–4,11]
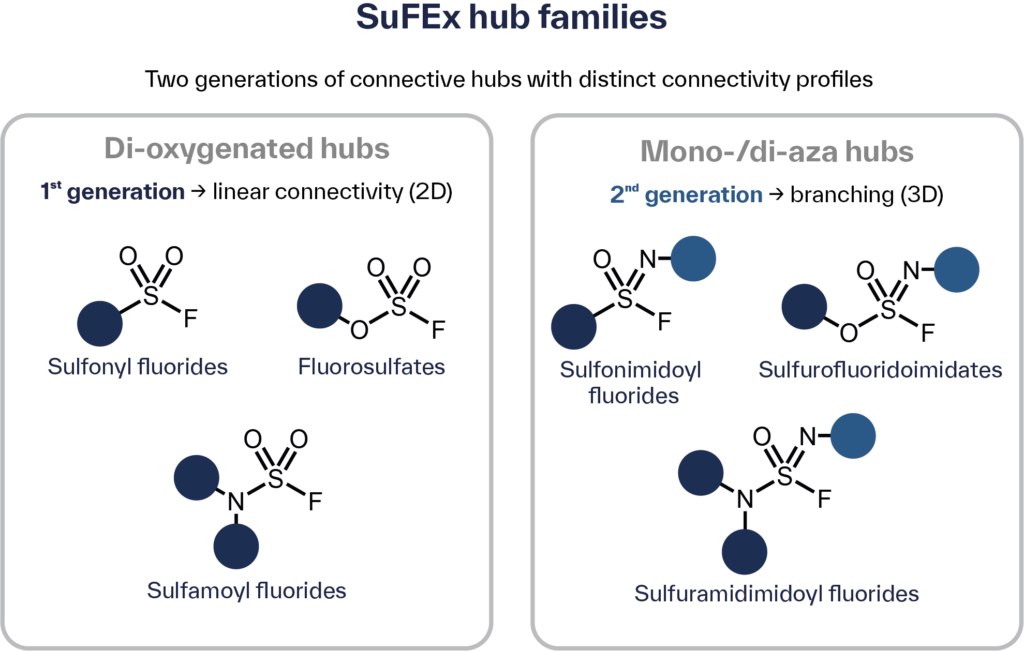
Figure 1: SuFEx handles and their connectivity profiles.
The Platform: SuFEx Hub Chemistry Meets AI-Driven Discovery
The platform brings together Melius Organics’ SuFEx hub technology, covering both di-oxygenated and the emerging mono-/di-aza families, with four REAXENSE discovery modules:
- ChemoVista™ gives immediate access to 11M+ curated, ready-to-ship compounds with property- and scaffold-level navigation, built for rapid hit identification and SAR follow-up.
- VirtuSynthium™ pushes the search space further, exploring ~10¹⁶ synthesis-ready virtual and generative molecules with real-time feasibility scoring.
- MacroCyclePro™ brings dedicated macrocycle design and optimization into the workflow, including bRo5-relevant design rules, with every series linked to a validated synthetic route.
- Receptor360™ makes the whole platform target-aware. It handles AI-driven protein characterization, conformational ensemble modeling, and automated pocket discovery (cryptic and allosteric sites included), then feeds directly into virtual screening and hit prioritization across the other three modules.
Together, these tools form an iterative, closed-loop workflow: target and pocket analysis feeds into SuFEx-aware library design and prioritization, which connects to synthesis and assay feedback, all within a single coordinated system (Figure 2).[5] On the synthesis side, the platform supports the full range of library strategies: from small, cherry-picked compound sets and focused selections through to high-throughput and Direct-to-Biology (D2B) plate-friendly formats with in situ screening workflows that cut purification bottlenecks and accelerate SAR cycles.[2,8,9]
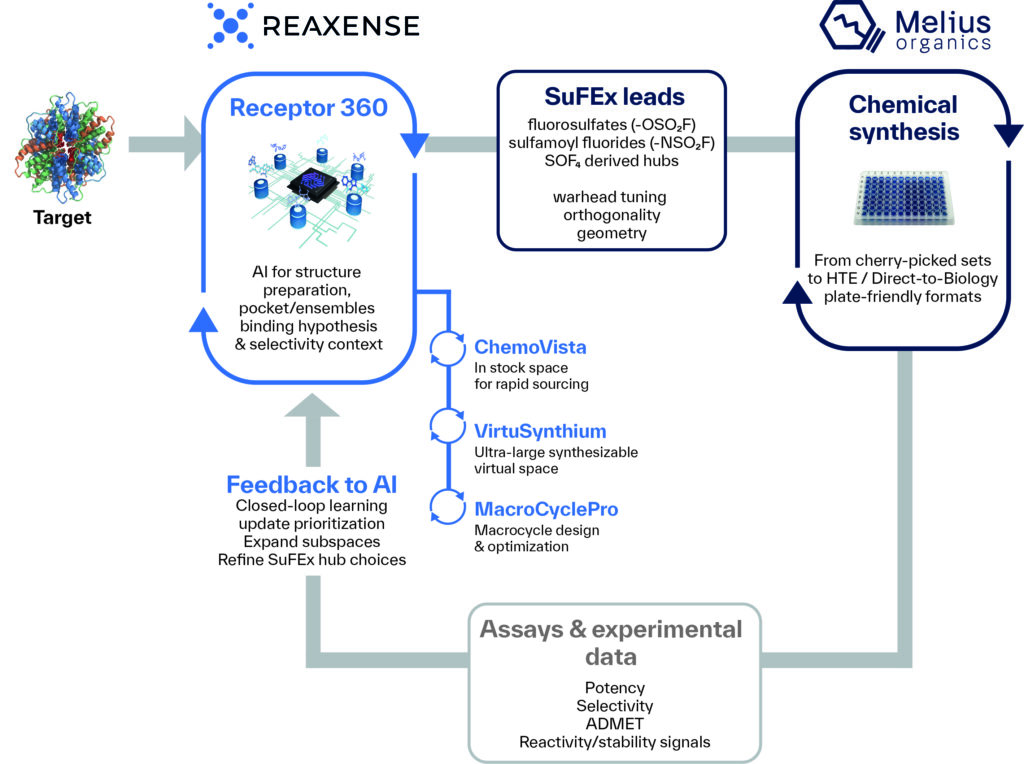
Figure 2: SuFEx Platform Workflow Diagram
Scientific and Practical Impact
The combination of SuFEx hub chemistry and AI-assisted design should compress the path from initial hits to optimized candidates. Modular, functionally tolerant library assembly means fewer synthetic dead ends and faster iteration.[2,11] The advantage comes from coupling inherently modular S(VI)–X linkages with makeability-aware virtual design and tight feedback between synthesis and assay data, so that prioritized compounds are both informative and synthesis-ready from the start.
Access to the newer mono- and di-aza hubs (e.g., SOF₄-derived iminosulfur oxydifluorides) gives medicinal chemists additional levers for controlling branching, vector orientation, and selectivity.[3,4] The platform also targets scalable routes to covalent fragments and covalent-ready libraries, with the aim of broadening S(VI)-based covalent inhibition beyond classical cysteine-targeting approaches.[10]
About Melius Organics
Melius Organics is a technology-driven chemistry company offering custom synthesis, CRO research, and scientific consulting for the pharmaceutical, agrochemical, and analytical sectors. Based in Reus, Spain, the company specializes in fluorine and sulfur chemistry, carbohydrate and amino acid synthesis, flow chemistry, and SuFEx hub technologies. Melius Organics also develops and supplies its own catalog of reagents and building blocks for drug discovery and related applications.
About REAXENSE
REAXENSE provides an AI-enabled chemistry and protein-centric discovery platform. It unifies curated in-stock compound space (ChemoVista™), ultra-large synthesis-ready virtual space generation (VirtuSynthium™), and modality-focused design engines such as MacroCyclePro™ with structure-guided target characterization (Receptor360™). Together, these modules support a closed-loop workflow, from target setup and pocket/ensemble analysis through virtual screening, prioritization, and synthesis-ready recommendations, so that teams can move faster without stitching together disconnected tools.
Media and Collaboration Inquiries
For collaboration discussions related to the SuFEx Platform, SuFEx Handle Fragment Library, or AI-enabled chemical-space design, please contact:
contact@meliusorganics.com | info@reaxense.com
References
- Dong, J. et al. (2014). Sulfur(VI) fluoride exchange (SuFEx): Another good reaction for click chemistry. Chem. Int. Ed. 53, 9430–9448.
- Homer, J. A. et al. (2024). Modular synthesis of functional libraries by accelerated SuFEx click chemistry. Sci. 15, 3879–3892.
- Li, S. et al. (2017). Multidimensional SuFEx click chemistry: Sequential sulfur(VI) fluoride exchange connections of SOF₄ Angew. Chem. Int. Ed. 56, 2903–2908.
- Liu, F. et al. (2019). Biocompatible SuFEx click chemistry: Thionyl tetrafluoride (SOF₄)-derived connective hubs for bioconjugation. Chem. Int. Ed. 58, 8029–8033.
- REAXENSE Inc. (2025). Platform description v4 (ChemoVista™, VirtuSynthium™, MacroCyclePro™).
- Melius Organics & OTAVA Ltd. (2025). SuFEx Handle Fragment Library brochure.
- Melius Organics (2025). SuFEx hub comparison (SO₂F₂, SOF₄, -OSO₂F, -SO₂F).
- Dada, L. et al. (2025). SuFEx-enabled high-throughput medicinal chemistry for developing potent tamoxifen analogs as Ebola virus entry inhibitors. Immunol. 16, 1533037.
- Kitamura, S. et al. (2024). Ultrapotent influenza hemagglutinin fusion inhibitors developed through SuFEx-enabled high-throughput medicinal chemistry. PNAS 121, e2310677121.
- Hansen, T. N. et al. (2025). Advances in sulfur fluoride exchange for chemical biology. Trends Chem. 7, 85–98.
- Liu, Z. et al. (2018). SuFEx click chemistry enabled late-stage drug functionalization. Am. Chem. Soc. 140, 2919–2925.






